|
|
|
|
|
![]()
Introduction ;
Ferrocene, namely cyolopentadienyl iron with the chemical
formula of Fe(C5H5)2, is an efficient and versatile additive and chemical
reagent. Ferrocene is a metallorganic complex with the smell of camphor. This
orange crystal has a melting point of 172-174¡æ, boiling point of 249¡æ. It
is dissolved in organic solvents such as benzene, diethyl ether, methanol, ethyl
alcohol, gasoline, diesel oil, and kerosene, but not in aqueous. It is
chemically steady and poisonousless, not reacting with acid, alkail and
ultraviolet. It doesn't decompose until 400¡æ. Mixed with Ferrocene, the
diesel oil can be preserved for a long use.
Quality Criterion:
|
Standard
|
Class
|
||
|
first |
second |
third |
|
|
Content (%) |
99 |
98 |
95 |
|
Precipitace content (%) |
0.1 |
0.2 |
0.3 |
|
Melting point (¡æ) |
172-174 |
||
|
Fe content (ppm) |
200 |
||
|
Organic soluent content (%) |
0.5 |
2.0 |
4.0 |
Application:
|
Used ad fuel catalyst for
rocket (aeroplane) propellant, it can improve combustion speed by 1-4
times, lower the temperature of exhaust pipes, and avoid infrared chase. |
||
|
It can be used as gasoline
antiknock (in place of tetrasthyl lead) to produce leadless gasoline. |
||
|
Used in fuel oils such as
diesel oil, heavy oil, light oil ect, It can eliminate smoke, save energy
and reduce air pollution. Adding 0.1% Ferrocene to diesel oil can cut down
the consumption of oil by 10--14%, eliminate smoke by 30--70%, and improve
power by more than 10%. And no deposit is left at the spray. So it plays a
dominant role in the development of fuel oils and improvement of their
moderate conditions. |
||
|
It can promote ammonia
synthesis reaction under moderate condition. |
||
|
As protective agent and
stabilizer, it can increase softening point and heat stability of polymer,
improve the properties of plastic, fuser, cellulose ect; protect
polynthene, polypropene, polyster ect, from depolymerization caused by
ultraviolet. |
||
|
Used in luminous sensitive
materials, it can replace silver on film. moreover, it can also be used in
photosensitive copy and printing with high resolution. |
||
|
It can be used for making large
scale integrated circuit board, enhancing light sensitivity by four times,
improving accuracy, simplifying technical process, and eliminating
pollution. |
||
|
As photosensitive catalyst, it
can promote the photochemical degadation of plastic films, reducing white
pollution. |
||
|
It is used as medical
ingredients for anticancer, antibiotic, and blood-tonic preparations. |
||
|
It can be used for producing
fertilizer. |
||
|
It can be used for making quality carbonicellulose. It can be used as biochemical and analytical reagent. |
![]()
Ferrocene's derivaties
Product List of Ferrocene Derivatives
1. acetyl ferrocene
2. propioly ferrocene
3. butyryl ferrocene
4. pentanoyl ferrocene
5. hexanoyl ferrocene
6. octanoyl ferrocene
7. benzoyl ferrocene
8. 1,1'diacetyl ferrocene
9. 1,1'-dibutyryl ferrocene
10. 1,1'-dihexanoyl ferrocene
11. ethyl ferrocene
12. propyl ferrocene
13. n-butyl ferrocene
14. pentyl ferrocene
15. hexyl ferrocene
16. 1,1'-diethyl ferrocene
17. 1,1'-dipropyl ferrocene
18. 1,1'-dibutyl ferrocene
19. 1,1'-dihexyl ferrocene
20. cyclopentenyl ferrocene
21. cyclohexenyl ferrocene
22. 3-ferrocenoyl propionic acid
23. 4-ferrocenoyl butyric acid
24. 4-ferrocenylbutyric acid
25. 5-ferrocenylvaleric acid
26. 3-ferrocenoyl propionic acid esters
27. 4-ferrocenoyl butyric acid esters
28. 4-ferrocenyl butyric acid esters
29. 5-ferrocenylvaleric acid esters
30. dimethylaminomethyl ferrocene
Products Introduction of Ferrocene Derivatives
The ferrocene derivatives become
commodity gradually at present years. Traditionally it is used as combustion
improved agent for the solid propellant of hydroxyl-terminated polybutadiene
binder. Which contributes greatly to its fast development. These products are
mainly used in the propelling system of rocket, missile and the other systems in
which the high burning rate is necessary. At present, the uses of these products
are increasing in various fields, such as petroleum, Plastic, textile,
metallurgy, mainly used as ultraviolet sensitizer, petroleum additives, dyes
additives, intermediate for production of metallocene, etc.
The quality of the serial products produced by us meet the standard stated in
some authoriarian chemicals catalogs in the world. The prices of our products
might certify our customers get more benefit, including the consumer or the
dealer.
![]()
|
|
Brief
Introduction;
Catocene is a new combustion improved agent of excellent
comprehensive properties developed for the adhexives solid propellant, for its
outstanding capability of combustion improvement and pressure reduction. Endued
with high boiling point, heat stability and Fe content as well as compatibility
to hydrocarbon adhesives, it is widely used in fields requiring combustion
enhancement and plastic decomposition in recent years.
Asides being a high-powered fuel itself, catocene is also an effective
functional auxiliary to petroleum. For instance, even 0.2% (weight-base) of
catocene is able to heighten the explosive force and driving safety of cars to
great extent so it becomes much popular in racing industry overseas lately. At
present catocene may be the only energy resolution sourcable to the problem
" moment speedup" from any aspect such as economic, scientific or any
other.
Moreover Catocene is also an eminent agent to plastic decomposition which
contributes a lot to garbage deposal and anti environmental pollution.
Quality description Technical description of Catocene (Q/GL 2-06-92)
|
Item |
Maximum |
Minimum |
|
Fe content (%) |
24.2 |
23.2 |
|
Purity (%) |
--- |
97.0 |
|
Viscosity (l¡¤pa) |
2500 |
--- |
|
Appearance |
brown viscous liquid |
|
America Military Standard MIL-C-85493 (AS) could be referred to.
Packing
500g or below, 1000g : in aluminum jar.
5000g, 10000g, 10kg or above : in plastic drum.
![]()
|
|
|
Series No. |
Colors |
|
PL Series |
Yellow-green, blue-green, green |
|
S Series |
Blue, blue-green, green,yellow |
|
R series |
red, orange-red, orange |
|
M series |
Blue, green, yellow, red. |
|
Paint, ink, ceramic glaze, resin series |
|

|
|
Caprolactam synthesis Introduction;
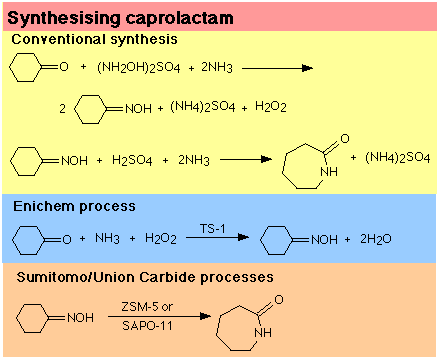
Caprolactam is a key raw material in nylon-6 manufacture. The conventional synthetic route to caprolactam is shown in Figure 3. Reacting cyclohexanone with hydroxylamine (produced as a sulphate salt by the air oxidation of ammonia to nitric acid, followed by catalytic hydrogenation in the presence of sulphuric acid) and ammonia yields an intermediate, cyclohexanone oxime. This undergoes a Beckmann rearrangement to caprolactam in the presence of stoichiometric amounts of sulphuric acid. Large quantities of salt are produced in both reactions (about 4.5kg of ammonium sulphate salt is generated per kilogram of caprolactam produced).
An alternative route to cyclohexanone oxime developed by Enichem in Italy is shown in Figure 3.33 The oxime is made by the ammoxidation of cyclohexanone with ammonia and aqueous hydrogen peroxide in the presence of a solid, recyclable catalyst, titanium silicalite (TS-1). This route eliminates about a third of the total salt formation. However, the oxime is still converted to caprolactam using the Beckmann rearrangement, producing ammonium sulphate salt. Thus, the process is still not as environmentally friendly as it could be.
A more suitable catalyst for converting cyclohexanone oxime to caprolactam is needed, in order to eliminate the salt formation. Sumitomo in Japan recently reported the use of a solid high-silica zeolite catalyst (ZSM-5) for the gas-phase rearrangement of cyclohexanone oxime at 350C.34 Caprolactam is produced with 95% selectivity at 100% oxime conversion. Union Carbide has also described the use of a proprietary catalyst, SAPO-11, for the same reaction.13 It is clear that application of these catalytic technologies can reduce unwanted salt generation, and replace sulphuric acid in these processes (see Figure 3).
CAPROLACTAM-WIDELY USED IN
THE AUTOMOBILE INDUSTRY
Caprolactam uses in the automobile industry have been increasing very rapidly as nylon components are being used as part of automobile engines. With the massive heat generated during the operation of vehicle engines, metal parts are subject to a great deal of wear, and the constant heating and cooling often causes distortion and damage over time. Nylon is both heat resistant, and a form of plastic which is more adaptable to these changes. Caprolactam is used in different nylon autoparts, such as various oil containers under the hood, wire harness connectors, fuse boxes, body frames for motorcycles, as well as covers for engine cylinders, crankcases, timing belts and wheels. Caprolactam is also used in making nylon tyre cords to increase the strength of automobile tyres.
Caprolactam’s main use is as the raw material for the production of nylon fibre, and consequently forms an integral part in a myriad of other consumer and industrial products outside the automobile industry.
INDUSTRIAL USE: Given its impact resistant, heat resistant, and excellent mold ability properties, nylon parts made of caprolactam are used in a wide range of industrial applications, including connectors for appliances, switch cases, coil bobbins, lamp sockets, and bodies and covers for engines. Its toughness and high weather ability also make nylon an ideal material for the production of deep sea fishing nets.
PACKAGING APPLICATIONS: Nylon film produced by caprolactam has excellent slipping properties with high transparency and low oxygen permeability. These characteristics make nylon film perfect for food packaging and liners for other packaging materials. As nylon is chemical resistant, nylon plastic bottles are also widely used as containers for agricultural chemicals.
TEXTILES: In the form of general textiles caprolactam is processed into many nylon materials, such as sports wear, swim wear, hosieries, buttons, carpets and fabric for tent, umbrella and bag.
FERTILIZER: The ammonium sulfate co-product of the caprolactam production process is used widely as fertiliser. Ammonium sulfate can be applied directly for crops such as rice, corn and sugar cane, or can be used as a key mix for NPK fertiliser. Thai Caprolactam Public Company Limited is the first producer of ammonium sulfate in Thailand where demand for ammonium sulfate of 600,000 tinners per annum had been met entirely by imports.
1. The polymer made from caprolactam is best described as a polyamide
2. The polymer most likely to have been made from a chain polymerization is Teflon & Plexiglass
3. Benzoyl peroxide is often used as an initiator for radical polymerization
4. Which of the following is NOT a major difference between low-density and high-density polyethylene?
e) C/H ratio
5. Which phrase best describes "living" polymerizations? polymerizations conducted in a manner such that chain-transfer and chain-termination steps are not significant.
Millions of pounds per year. AlliedSignal uses phenol as a raw material, while BASF and DSM use cyclohexane. AlliedSignal and BASF consume most of their monomer production captively in their respective nylon operations. DSM is a merchant supplier, but it will add a significant captive requirement for caprolactam once a world-scale nylon 6 polymerization plant the company is building at Augusta comes on line in the fourth quarter of this year. DSM, which intends to remain a merchant supplier, has also formed a 50-50 joint venture with AlliedSignal to depolymerize nylon 6 to caprolactam from used carpet. The companies will divide the 100 million pounds of merchant-grade caprolactam the venture will produce each year when the plant reaches full production. Plans call for construction of an $80 million manufacturing facility at the Augusta site to begin this year, with production due in mid-1999. BASF has been operating a recycling program for its nylon 6 since 1994. The company plans a capro debottlenecking to add 45 million pounds of capacity. Univex, in which a partnership of DuPont and Mexico's Alpek SA has a majority stake, operates a 165-million-pound caprolactam facility at Salamanca. Profile last published 10/2/95; this revision 3/2/98.
DEMAND
1997: 1.57 billion pounds;
1998: 1.60 billion pounds;
2002: 1.76 billion pounds (Includes exports, which amounted to 183 million
pounds in 1996 and averaged 184 million pounds annually in the 1992-1996 period.
Imports totaled 56 million pounds in 1996 and averaged 34 million pounds a year
during the period.
GROWTH
Historical (1988-1997): 2.3 percent per year;
future: 2 percent per year through 2002.
PRICE
Historical (1981-1995): High, 96c. per pound, list,
molten, tanks, f.o.b. shipping point; low, 74.5c. per pound, same basis.
Current: 93c. per pound, same basis.
USES
Nylon 6 fibers, including monofilament, 80 percent;
engineering resins and film, 20 percent.
STRENGTH
Nylon is expected to continue to dominate the
3.5-billion-pound synthetic carpet fibers market, based on price/performance
balance and mill processing attributes. Growth in carpet and rug markets have
offset decline in apparel. Nylon 6 fiber, while growing at less than 2 percent
annually, will still require an estimated 1.2 billion pounds of caprolactam at
the turn of the century. Increased application of nylon 6 resins in automotive
and truck parts has led to double-digit growth in the segment in recent years.
WEAKNESS
Nylon has sustained losses in tire cord because of
market penetration by steel-belted radial tires. Nylons resins must compete with
acetals and polycarbonate in engineering thermoplastics applications.
OUTLOOK
Stable fiber demand is projected through the end of
the decade, while above-average growth in high-end engineering resins will
provide most of the momentum for nylon 6 and caprolactam markets. Capro's
ongoing snug supply/demand balance would seem to warrant new construction, but
grassroots projects are costly, and producers have chosen to stay with extensive
debottlenecking programs. Also, commercialization of processes for
depolymerization of used nylon 6 promise an additional low-cost source of
feedstock caprolactam.
Conclusions
While safer chemistry practices are occurring, they are often a by-product of other goals, such as pollution prevention or economics. With increasing government and public interest in chemical safety following the 1984 accident in Bhopal, the identification of inherently safer chemical processes has begun to accelerate, and should be encouraged, on the merits of the processes themselves. Indeed, the ultimate cost to industry may be high if it does not adequately address potential chemical risks that later become actual emergencies, with their resulting consequences. It is hoped that this article will stimulate thinking in the direction of inherently safer chemistry. The case studies mentioned here represent only the 'tip of the iceberg', because they are only a few of the hundreds of industrial chemical processes. Their extrapolation potential to other similar types of chemistry has yet to be realized.
In case of further information
needed, Please feel free to advise to us. Thank You,
